Build Your Geriatric Assessment Program Blueprint
Apr 20, 2026

Teams exploring a geriatric assessment program are often already feeling the strain. The older adult in front of them isn’t just managing hypertension or arthritis. They may also be forgetting appointments, walking more slowly, eating less, sleeping poorly, and relying on a daughter who’s close to burnout. A routine visit catches fragments of that picture. It rarely captures the whole pattern.
That gap matters. If you miss the interaction between mobility decline, mood change, medication burden, and early cognitive impairment, you don’t just miss diagnoses. You miss the care plan that would help.
A well-built geriatric assessment program gives clinicians a way to organise complexity instead of reacting to it piecemeal. It creates a repeatable process for identifying risk, assigning the right disciplines, and converting findings into action. Done well, it also becomes operationally sustainable. That’s where many programmes either mature or stall.
Beyond the Annual Physical Understanding the Need for a Geriatric Assessment
An annual physical is useful. It checks blood pressure, reviews preventive care, updates medications, and may cover a few screening questions. But it usually isn’t built to answer the questions that dominate late-life care.
Can this person still manage meals, bills, and transport safely? Is their slower gait a deconditioning problem, a neurological one, or both? Is low mood worsening adherence? Is the family support system strong enough to keep the patient at home?

What a geriatric assessment program does differently
A geriatric assessment program is a structured, multidimensional review of health, function, cognition, mood, medications, and social context. It doesn’t treat ageing as a single diagnosis. It looks for interacting drivers of decline.
That distinction is important in frailty care. Older adults often present with syndromes rather than neat disease categories. Falls, weight loss, confusion, incontinence, caregiver strain, and loss of independence tend to travel together. A programme approach helps a team sort which issues are urgent, which are reversible, and which require ongoing support.
A useful overview of that broader framework appears in this guide to comprehensive geriatric assessment in Canada.
What the evidence says, and what it doesn’t
Programmes like this shouldn’t be sold as magic. A systematic review of 7 randomized clinical trials involving 3,254 subjects aged 60+ found no significant impact on mortality or several other hard endpoints versus usual care, though it did find favourable quality of life improvements at 3 and 12 months based on very low-quality evidence in individual studies, according to this systematic review of outpatient comprehensive geriatric assessment.
Practical rule: Build a geriatric assessment program to improve decision-making, coordination, and day-to-day wellbeing. Don’t promise mortality gains the evidence doesn’t support.
That’s the right lens. In practice, the value of a GAP often shows up in clearer care plans, better discharge decisions, smarter referrals, and fewer avoidable surprises for families and staff.
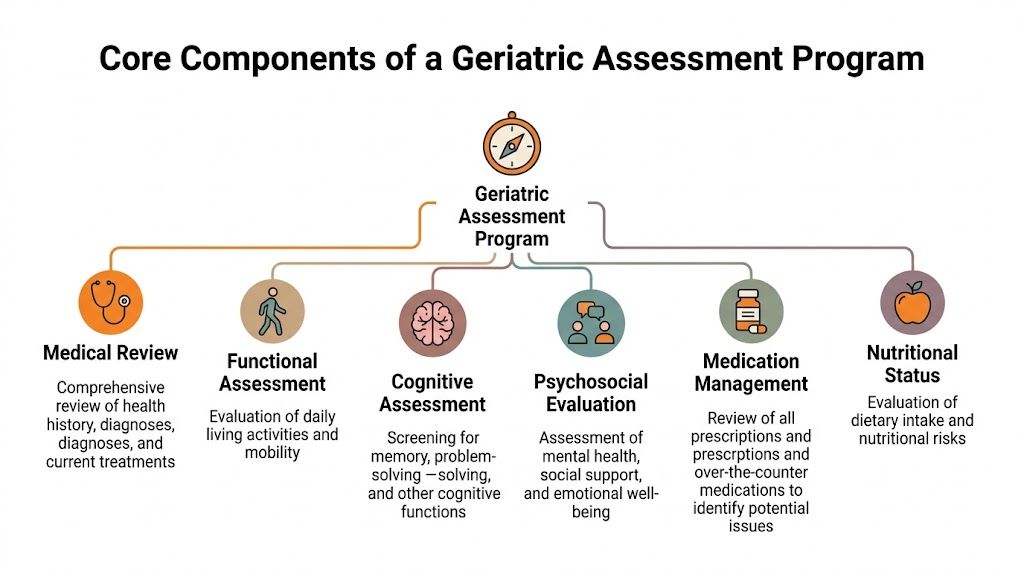
The Core Components of a Geriatric Assessment Program
A standard annual visit is like an oil change. A geriatric assessment program is closer to a full vehicle diagnostic. You’re not only checking whether the engine turns on. You’re looking at brakes, steering, warning lights, tyre wear, fluid leaks, and whether the driver can still operate the car safely.
That depth only works when the programme has two solid pillars. First, a team that knows how to look at the patient from different angles. Second, a domain structure that ensures nobody forgets the issues that most often drive decline.
The multidisciplinary team
A strong GAP doesn’t need every specialist in the building at all times. It does need role clarity.
Geriatrician or lead physician handles diagnostic integration, prognosis discussions, medical prioritisation, and decisions about what should be treated aggressively versus monitored.
Nurse often runs intake, screening, vitals, history refinement, and follow-up coordination. In many programmes, the nurse is the operational backbone.
Pharmacist reviews prescriptions, over-the-counter drugs, duplicate therapies, adherence patterns, and side-effect burden.
Social worker identifies caregiver capacity, housing concerns, financial strain, access barriers, and community resource needs.
Physiotherapist or occupational therapist assesses gait, transfers, balance, home safety, and the person’s actual ability to manage daily tasks.
Psychologist or neuropsychology-trained clinician helps clarify whether cognitive complaints reflect mild impairment, depression, delirium, language barriers, low literacy, or a more complex neurocognitive disorder.
If you need a quick refresher on daily functioning categories, this breakdown of ADL vs IADL is useful when designing forms and referral triggers.
The assessment domains
Most programmes become inconsistent when they rely too heavily on clinician memory. Domain-based design fixes that. Every patient should move through a stable set of domains, even if the depth varies.
Domain | What the team looks for | Practical example |
|---|---|---|
Medical review | Diagnoses, symptom burden, comorbidities, treatment burden | A patient’s fatigue may reflect heart failure, anaemia, sedating medication, or depression |
Functional ability | Basic and instrumental daily tasks | Difficulty with shopping and finances may appear before trouble with dressing or bathing |
Cognition | Memory, attention, executive function, language, processing | Missed pills may be a cognition problem, not “non-compliance” |
Psychological health | Mood, anxiety, adjustment, grief, motivation | Low mood can mimic or worsen cognitive symptoms |
Social and environmental context | Caregiver support, transport, language, housing, isolation | A good plan fails if the patient can’t get to therapy or has nobody to supervise medications |
Medication management | Polypharmacy, interactions, complexity | A fragmented regimen can cause dizziness, confusion, and falls |
What works and what doesn’t
Teams get better results when they stop treating the assessment as a form-filling exercise and start treating it as a decision system.
What works:
Structured intake templates
Defined referral thresholds
A single care-plan summary
Case conferences with accountable owners
What doesn’t:
Collecting data nobody uses
Running separate assessments that never reconcile
Overbuilding the team before patient flow exists
Using long test batteries for every patient, regardless of need
The point isn’t maximum detail. It’s clinically useful detail.
Anatomy of an Assessment Workflow and Essential Tools
The easiest way to test a geriatric assessment program is to follow one patient through it.
An older adult is referred after two falls, missed medications, and concerns from family about forgetfulness. Intake starts before the face-to-face visit if possible. Staff gather the medication list, referral reason, recent admissions, mobility aids, and the family member most involved in care. That alone often exposes the first operational problem: the chart says “independent”, while the daughter says she now handles meals and banking.
Step one through step three
At the first encounter, the nurse or lead assessor usually does three jobs in sequence.
Triage urgency Decide whether this is a stable outpatient review or whether delirium, acute decline, or safety concerns need immediate escalation.
Screen the key domains Mobility, function, mood, cognition, medications, supports, and recent change all need early attention.
Route the patient Not everyone needs every discipline on day one. The programme should decide who gets same-day therapy input, medication review, social work, or deeper cognitive testing.
A practical cognitive overview can start with standard screeners and then move to more detailed tools when the picture is unclear. This summary of cognitive screening tests for dementia is helpful when building that escalation pathway.
Mobility and fall risk tools
Mobility testing is one of the fastest ways to uncover hidden risk. The Timed Up and Go, or TUG, is a core example because it reflects transfers, gait, turning, and balance in one simple task.
A TUG time above 12 seconds indicates high fall risk, and evidence cited in the Merck Manual notes that targeted interventions based on TUG findings can reduce fall incidence by 25 to 30%, as outlined in this Merck Manual review of comprehensive geriatric assessment.
That matters operationally. A slow TUG should trigger more than a note in the chart. It should drive action such as physiotherapy referral, gait aid review, home safety planning, and medication scrutiny for sedatives or hypotensive agents.
A typical mobility sequence may include:
TUG first: Fast, repeatable, and useful at intake.
Observed transfers: Can the patient stand safely without using momentum or pulling on furniture?
Gait observation: Watch stride length, hesitancy, turning, and dual-task difficulty.
Context questions: Ask where falls happen, not just how many occurred.
Mood, function, and cognition
Mood and cognition often overlap in older adults. If you screen one and ignore the other, you can head in the wrong clinical direction.
The Geriatric Depression Scale is commonly used because it’s practical and acceptable for older adults in many settings. Functional review should look at both ADLs and IADLs. A patient may still dress independently while losing the ability to organise medications, prepare meals, or manage money.
For cognition, many programmes use tools such as the MoCA or MMSE depending on training, workflow, and patient factors. The key isn’t loyalty to one instrument. The key is consistency, proper interpretation, and a plan for what happens when results are borderline, language-dependent, or discordant with real-world function.
A cognitive score without functional context is incomplete. Functional decline without cognitive review is also incomplete.
The team conference and final care plan
The most important meeting in the programme often happens after the patient leaves the room. The disciplines compare findings and build one plan. Not six parallel plans.
A useful case conference usually answers these questions:
What are the top problems? Limit to the issues that most affect safety, function, and quality of life.
What is reversible? Medication side effects, untreated depression, deconditioning, sensory issues, poor sleep, and caregiver overload often sit in this category.
Who owns each action? Assign a clinician, deadline, and follow-up point.
What does the family need to understand today? Not every detail. Only the details that change care.
Weak programmes generate long reports. Effective ones generate clear next steps.
Your Implementation Roadmap Building a Program from the Ground Up
Most organisations don’t fail because the clinical idea is weak. They fail because the launch is vague. A geriatric assessment program needs a build plan with staffing decisions, referral rules, documentation standards, and a way to prove value early.

Phase one, secure buy-in with an operational case
Leaders rarely approve programmes because the concept sounds admirable. They approve them when the proposal solves visible problems.
Frame the case around issues administrators already recognise:
delayed discharges
repeat falls
preventable readmissions
medication-related complications
caregiver complaints
inefficient specialist referrals
Feasibility matters here. In an Australian transition care study, the full suite of assessments took an average of 55.9 ± 7.3 minutes, excluding MoCA, with 100% completion for most tools and 90% usability for the 10m Walk Test, according to this study of comprehensive geriatric assessment feasibility. That’s useful when someone says, “This sounds too heavy for clinic flow.”
Phase two, choose a staffing model that fits reality
Not every site can hire a full dedicated team at launch. That’s fine. Build the model that your referral volume and budget can support.
Model | Best fit | Main trade-off |
|---|---|---|
Dedicated clinic team | Larger hospitals, rehab centres, high referral volume | Better consistency, higher fixed staffing cost |
Consultative network | Community settings, smaller systems | More flexible, but coordination can drift |
Hybrid model | Most programmes starting out | Strong core team with specialist consults as needed |
A practical starting configuration is often a nurse-led intake process, physician oversight, pharmacist access, and therapy referral pathways. Social work can be embedded or consult-based depending on local need.
For teams building around discharge support and longitudinal management, continuity planning matters as much as the initial assessment. This guide to continuity of care can help shape handoffs and follow-up rules.
Phase three, build workflow before volume rises
Many programmes become chaotic. Don’t wait for demand to expose process gaps.
Use a simple implementation checklist:
Define referral criteria: Falls, functional decline, memory concerns, polypharmacy, repeated admissions, caregiver stress.
Standardise intake packets: Medication list, recent events, current supports, reason for referral.
Create one shared documentation template: Every discipline should feed into one summary.
Set conference cadence: Same day, weekly, or twice weekly depending on volume.
Assign communication responsibility: Decide who calls the family, who updates the primary clinician, and who books follow-up.
Build escalation rules: Delirium, safeguarding concerns, unsafe home return, severe depression, medication toxicity.
Phase four, track success with the right measures
Avoid vanity metrics. Counting referrals alone won’t protect the programme.
Track measures that change decisions:
Completion rate of core assessments
Time from referral to assessment
Rate of actionable medication changes
Therapy referrals completed
Functional change over follow-up
Hospital use or readmission trends
Care-plan completion and handoff quality
Field note: Start with a small metric set you’ll actually review every month. Teams abandon dashboards that try to measure everything.
When the first six months are messy, that isn’t proof the programme is failing. It usually means your referral criteria are broad, your documentation is fragmented, or your follow-up ownership isn’t clear. Fix those first.
Modernizing GAPs with Digital Cognitive Assessment Platforms
Traditional cognitive testing still has a place, but it also has limits that become obvious the moment a programme tries to scale. Paper tools can be slow to score, sensitive to administrator technique, hard to trend over time, and awkward to integrate into a busy clinic record. They also tend to compress complex cognition into a few summary points.
That’s not enough for many real-world geriatric cases. Clinicians often need more than “screen positive” or “screen negative.” They need a clearer profile. Is the main issue attention, memory encoding, executive control, processing speed, or a mixed pattern? If the answer is vague, the intervention plan stays vague too.
Why digital assessment changes the workflow
A major gap in geriatric care is scalable cognitive assessment, especially in settings with specialist shortages. One review focused on California noted that AI-powered tools can provide a detailed cognitive profile in under 30 minutes and may support personalised care plans, with broader CGA studies suggesting potential to reduce hospital readmissions by up to 20%, as discussed in this analysis of scalable cognitive assessment in ageing populations.
The practical advantage isn’t just speed. It’s structure.
Digital cognitive platforms can help a GAP by:
Standardising administration across staff and locations
Producing objective domain-level data instead of relying only on subjective impressions
Supporting repeat measurement so decline or improvement is easier to track
Reducing scoring lag between assessment and care-plan discussion
Improving triage for who needs deeper neuropsychology or neurology follow-up
For organisations evaluating online options, this overview of cognitive assessment online is a practical starting point.
Where digital tools fit, and where they don’t
Digital tools work best when they extend clinical judgement, not replace it. A platform can improve precision around attention, memory, executive function, processing speed, perception, and eye-hand coordination. It can’t independently diagnose dementia, determine capacity in isolation, or explain every social factor behind poor performance.
That’s the trade-off to keep in view. Better data is only useful if the team knows how to interpret it in context.
One option in this space is Orange Neurosciences, which provides AI-powered cognitive assessment in under 30 minutes and generates objective profiles across multiple domains for care planning and longitudinal tracking. Used properly, that kind of platform can make a geriatric assessment program more scalable without turning it into a tech-first service.
A sensible integration model
The best implementation model is usually selective, not universal.
Use digital cognitive assessment when:
standard screening is inconclusive
the patient has subtle decline affecting IADLs
you need a more detailed baseline before rehab or monitoring
repeat reassessment is likely
specialist access is delayed
Keep traditional clinician-led methods front and centre when:
delirium is suspected
severe sensory or language barriers make testing invalid
the patient is too unwell or fatigued
the main issue is behavioural crisis or urgent safety planning
A modern programme doesn’t choose between traditional assessment and digital assessment. It uses each where it adds the most value.
Navigating Reimbursement Quality Measures and Ethical Considerations
The financial question arrives quickly. Can the programme sustain itself, especially if it relies on more than one discipline? In many places, reimbursement for assessment work trails far behind the staff time needed to do it properly.
In California, Medi-Cal may reimburse only $150 to $300 for a full geriatric assessment, which is often inadequate for a multidisciplinary model. At the same time, emerging telehealth and tech-enhanced programmes have reported cost savings of up to 35% by improving efficiency and scalability, according to this review of reimbursement and technology-enabled geriatric care models.
What that means in practice
That payment gap creates a simple operational truth. A GAP often has to justify itself through system performance, not just direct billing.
Administrators usually respond to arguments tied to:
reduced avoidable utilisation
better discharge planning
lower duplication across services
clearer documentation for follow-up care
improved fit with value-based care expectations
You don’t need heroic financial modelling to make the case. You do need honest accounting. If the programme requires physician time, nursing coordination, therapy input, and pharmacy review, the budget has to reflect that mix.
Quality measures and care standards
A good programme also helps with quality oversight because it makes risk visible earlier. Falls risk, medication burden, cognitive concerns, caregiver strain, and loss of function become structured findings rather than scattered chart comments.
That structure supports better handoffs into community services. When a patient’s needs move beyond clinic-based management, local home support with relevant expertise matters. For families trying to understand what specialised support can look like outside the clinic, these specialized dementia care services offer a useful example of how day-to-day support adapted to individual needs may complement formal assessment findings.
Ethical guardrails that shouldn’t be optional
Ethics in a geriatric assessment program isn’t a side topic. It sits inside routine operations.
Keep these standards explicit:
Consent: Explain what’s being assessed, why it matters, and who will see the results.
Privacy: Cognitive and psychosocial findings are sensitive. Access should follow role-based need, not curiosity.
Family communication boundaries: Involved relatives help care, but the older adult still deserves direct communication whenever possible.
Bias awareness: Language, education, hearing loss, culture, and digital familiarity can all distort assessment performance.
Use limitation: Assessment data should guide care. It shouldn’t be stretched beyond the purpose for which it was collected.
If a programme gathers sensitive data faster than it can protect, interpret, and explain it, the programme isn’t modern. It’s careless.
Conclusion Building the Future of Senior Care
A strong geriatric assessment program gives teams a practical way to manage complexity that routine visits often miss. It brings together medical review, function, mobility, mood, cognition, medication management, and social context. It then turns those findings into a single care plan with clear ownership.
The programmes that work best are disciplined about workflow. They define who gets referred, which tools are used, when the team meets, and how follow-up happens. They also stay realistic about trade-offs. Thorough doesn’t have to mean cumbersome, and digital doesn’t have to mean impersonal.
The next step for many organisations is modernisation. Digital cognitive assessment can make programmes more scalable, more consistent, and easier to track over time, especially where specialist access is tight and teams need better data without adding endless manual work.
If you’re building or upgrading a geriatric assessment program, explore how Orange Neurosciences can support cognitive profiling, reassessment, and care planning by visiting the website or contacting the team for a demonstration.
Frequently Asked Questions About Geriatric Assessment Programs
What’s the difference between a geriatric assessment and a Medicare Annual Wellness Visit
They serve different purposes. An annual wellness visit is primarily a preventive and review-focused encounter. A geriatric assessment is broader and more problem-solving in nature.
A proper geriatric assessment usually goes deeper into:
functional decline
falls and mobility
cognition and mood
medication burden
caregiver situation
home and social supports
If an older adult has recent decline, recurrent falls, confusion, or increasing dependence, a wellness visit alone usually won’t answer the clinical questions that matter most.
Can a smaller clinic or solo practice implement a lighter version
Yes. A smaller site can run a GAP-lite model if it stays structured.
Start with:
a defined referral trigger list
one functional screen
one mobility test
one mood screen
one cognitive screen
a medication review process
a referral network for therapy, pharmacy, and social work
What smaller clinics shouldn’t do is mimic a hospital programme on paper without the staff to carry it out. Keep the core consistent, then refer outward for deeper assessment when complexity exceeds local capacity.
What are the ethical guidelines for sharing findings with family and caregivers
Start with the older adult whenever possible. Explain results in plain language and ask who they want involved. Capacity concerns, safety risks, and local legal duties can change how information is shared, but family access shouldn’t be automatic just because they attended the visit.
Use a simple rule set:
Ask permission first when the patient can provide it
Share only what supports care
Separate clinical facts from speculation
Document who received the information and why
Escalate carefully if safety is at risk
How often should a geriatric assessment be repeated
There isn’t one universal schedule that fits every setting. Reassessment should follow clinical change, not just the calendar.
Repeat the assessment sooner when there’s:
a fall
a hospitalisation
a major medication change
clear functional decline
new cognitive concern
rising caregiver strain
Stable patients can often be reviewed at planned intervals set by the programme. The key is to reassess when the result will alter management.
If you’re looking for a practical way to modernise your geriatric assessment workflow, Orange Neurosciences offers AI-powered cognitive assessment and tracking tools that can fit into a broader care model. Visit the site or contact the team to see how digital cognitive profiling can support more precise, scalable senior care.

Orange Neurosciences' Cognitive Skills Assessments (CSA) are intended as an aid for assessing the cognitive well-being of an individual. In a clinical setting, the CSA results (when interpreted by a qualified healthcare provider) may be used as an aid in determining whether further cognitive evaluation is needed. Orange Neurosciences' brain training programs are designed to promote and encourage overall cognitive health. Orange Neurosciences does not offer any medical diagnosis or treatment of any medical disease or condition. Orange Neurosciences products may also be used for research purposes for any range of cognition-related assessments. If used for research purposes, all use of the product must comply with the appropriate human subjects' procedures as they exist within the researcher's institution and will be the researcher's responsibility. All such human subject protections shall be under the provisions of all applicable sections of the Code of Federal Regulations.
© 2026 by Orange Neurosciences Corporation


