Clinical Trials In Montreal: Find Studies & Participate
Apr 23, 2026

A lot of people start searching for clinical trials in montreal on a hard day.
A specialist has just said standard treatment may not be enough. A parent is trying to understand whether a research study could open another option for a child. A clinician is looking for something more targeted for a patient whose symptoms aren’t improving. The search usually begins with equal parts hope and confusion.
That confusion is understandable. Clinical trials can sound distant, technical, and full of gatekeepers. In reality, they’re often much more practical than people expect. They involve real hospitals, real coordinators, real scheduling calls, and real decisions about time, travel, consent, and whether a study is a good fit.
Montreal is one of the strongest places in Canada to explore that path. The city combines major hospital networks, specialised research institutes, bilingual care environments, and a culture of academic medicine that keeps new studies moving. That matters whether you’re looking for a cancer trial, a neurology study, a liver study, or a way to bring stronger cognitive measurement into research practice.
If you’re working in brain health, it also helps to see how digital tools are beginning to support care and research across the country, including projects such as this Canadian digital brain health expansion effort.
Exploring New Hope Through Research in Montreal
A family from Côte-des-Neiges comes to clinic after months of appointments. Their loved one has already tried the usual options. The doctor isn’t promising a miracle, but does mention a study at another Montreal hospital. Suddenly the conversation changes. Instead of asking only, “What has failed?” the family starts asking, “What’s available?”
That moment is common in hospital practice. Clinical trials often enter the picture when patients need another route, when clinicians want access to emerging therapies, or when researchers need better ways to measure whether an intervention is helping.
Montreal is especially well placed for that kind of next step. The city isn’t just large. It’s organised around teaching hospitals, university networks, and specialised centres that bring research close to patient care. For people searching for clinical trials in montreal, that means you’re not looking in a weak or isolated market. You’re looking in a city where research is part of everyday medicine.
Still, hope needs structure. Patients need to know how trials work, how to judge a listing, and what daily participation really feels like. Families need help separating a reasonable opportunity from a poor fit. Researchers need practical methods for recruitment, data collection, and follow-up that work in practice, not just on paper.
Clinical research is most useful when it becomes understandable. A study only helps if people can find it, evaluate it, and participate in it with confidence.
That’s where a practical guide matters. The goal isn’t to make every reader into a research expert. It’s to make the system less intimidating. Once people understand the rules, the hospitals, the search tools, and the common obstacles, clinical trials become easier to approach.
Understanding Clinical Trials in a Quebec Context
A clinical trial often enters a Montreal family’s life in a very practical way. A neurologist mentions a study visit. A daughter asks whether RAMQ covers routine care. A spouse wonders who will read the consent form in French or English, and whether the extra hospital trips are realistic in winter.
That is the Quebec context. A trial is never just a scientific protocol. It is also a schedule, a set of approvals, a travel plan, and a series of decisions that patients and care teams have to make under stress.
Clinical trials are organised studies that test medical approaches in people. The intervention might be a drug, a device, a rehabilitation program, a change in care delivery, or a new way to measure symptoms over time. The basic goal is simple: learn in a structured way, with safeguards, whether an approach is safe, useful, and practical.
One comparison helps. Trial development works like opening sections of a bridge in sequence. Engineers do not send full traffic across on day one. They test materials, load, and stability in stages. Clinical research follows the same logic. Early studies ask whether an intervention can be used safely. Later studies ask how well it works, for whom, and how it compares with current care.

The four phases in plain language
Phase labels sound technical, but the questions behind them are familiar.
Phase | Main question | What patients usually notice |
|---|---|---|
Phase 1 | Is the intervention safe enough to test in people, and what dose or usage pattern makes sense? | Closer monitoring, more safety checks, and a stronger focus on side effects |
Phase 2 | Does it show signs of benefit, and what is the best way to use it? | More attention to whether symptoms, biomarkers, or functional measures change |
Phase 3 | Does it perform well in a larger group when compared with standard care or another approach? | More standardised visits and clearer comparison between groups |
Phase 4 | What happens after approval in routine care? | Longer-term follow-up and monitoring in real-world use |
For patients, the phase affects the feel of participation. An early-phase oncology study may involve frequent bloodwork and careful dose review. A later-phase rehabilitation or neurology study may involve repeated assessments over months, with less focus on dose-finding and more focus on daily function.
For researchers, each phase changes the operational load. Consent tracking, protocol amendments, source documents, deviation logs, and visit windows all need careful handling. Teams reviewing their processes can use this overview of GxP documentation requirements to tighten recordkeeping before small errors become recruitment delays or audit findings.
What makes Quebec distinct
Quebec combines national regulation with local ethical review and local care realities. Health Canada may authorise a drug trial at the federal level. A hospital or university research ethics board, often called a Comité d’éthique de la recherche, reviews whether the study is acceptable for participants at the site level. Those are different jobs. One looks at the investigational product and regulatory submission. The other looks closely at consent, privacy, risk, recruitment, and how the study will run with real patients in a real institution.
That difference matters because approval on paper does not solve practical barriers on the ground.
In Montreal hospitals, families often need answers to questions that never appear in a trial abstract. How many visits are in person? Can someone accompany the participant? Are parking, transit, or interpreter needs considered? If a participant becomes tired, cognitively overloaded, or too unwell to finish a long assessment battery, is there a backup plan that still protects data quality?
These details shape participation as much as eligibility criteria do.
Quebec’s bilingual clinical environment adds another layer. Consent discussions, symptom reporting, and cognitive testing all depend on clear language. A participant who understands the treatment goal but struggles with the wording of an assessment may look worse on paper than they are in daily life. That is one reason research groups tied to Montreal academic institutions and clinical research partners increasingly pay attention to how assessments are delivered, not just which assessments are chosen.
A practical rule for patients, families, and clinicians
A legitimate trial site should be able to explain five things in plain language:
who reviewed and approved the study
what each visit involves
which costs are covered and which are not
what the known risks and uncertainties are
how personal health data and study data will be stored, shared, and monitored
If any of those answers are vague, ask again.
For clinicians referring patients, the same rule applies. A promising protocol is not enough. The study also needs realistic visit timing, a consent process suited to the patient population, and outcome measures that people can complete reliably. In neurology, geriatrics, psychiatry, and rehabilitation, that last point is often the weak spot. Long in-clinic cognitive testing can be tiring, hard to standardise across sites, and difficult for participants who depend on caregivers or public transit.
Modern cognitive assessment tools can reduce some of that burden. Used well, AI-supported testing and monitoring can help research teams collect more consistent data, spot changes earlier, and reduce unnecessary in-person strain on participants. For families, that can mean fewer exhausting visits. For researchers, it can mean cleaner follow-up data and fewer missing assessments.
Clinical trials in Quebec work best when the science and the lived reality line up. Patients need safeguards, clear explanations, and practical scheduling. Researchers need methods that fit hospital workflows and patient capacity. When both sides are considered together, participation becomes easier to judge and studies become stronger.
Montreal's Leading Clinical Research Hubs
A family in Montreal may see a specialist at one hospital, complete imaging at another, and hear about a study taking place somewhere else entirely. That can feel confusing at first. In practice, it reflects how research works across the city. Montreal’s clinical trial system is a connected network of hospitals, university institutes, specialty clinics, and research teams that each serve different needs.
For patients, this matters because the best trial site is not always the place where you usually receive care. For clinicians and study teams, it matters because referrals, testing, pharmacy support, and follow-up often depend on partnerships across institutions. The city’s research strength comes from that shared structure, not from any single building.
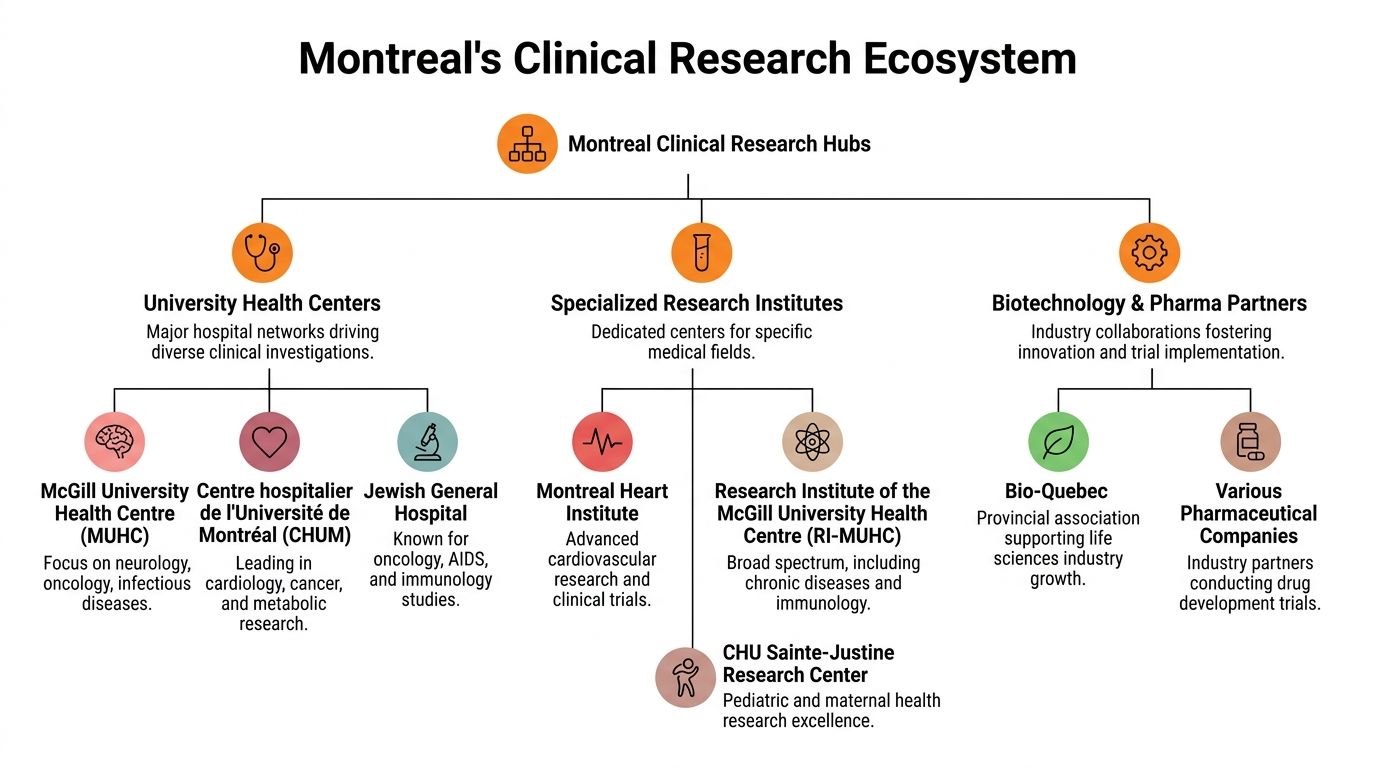
Jewish General Hospital and the Lady Davis Institute
The Jewish General Hospital (JGH) and the Lady Davis Institute for Medical Research (LDI) are well known in Montreal for active clinical research across multiple disease areas. A centre with that kind of trial experience usually has established systems for screening participants, coordinating ethics approvals, managing investigational medications, and keeping follow-up visits on schedule.
That operational side is easy to overlook, but it affects the patient experience directly. A study can look promising on paper and still be hard to complete if booking is disorganized or visits are too difficult to manage. Sites with experienced trial teams are often better at reducing that friction.
Within JGH, the Segal Cancer Centre is a major oncology research setting. Early-phase studies and later-stage cancer trials may run through different units or linked programs, so patients should ask exactly where the study visits occur, how often they are required, and whether bloodwork, imaging, or symptom tracking can be coordinated with routine care.
MUHC and the Research Institute of the MUHC
The McGill University Health Centre (MUHC) and the Research Institute of the MUHC are another key part of Montreal’s research system. Their strength often comes from cross-specialty work. That is especially helpful for people whose condition does not fit neatly into one clinic, such as a patient with neurological symptoms, psychiatric concerns, and rehabilitation needs at the same time.
For researchers, this kind of environment can support more realistic study design. If a protocol requires cognitive testing, imaging, specialist review, and longitudinal follow-up, the question is not only whether the science is sound. The question is whether participants can complete those steps without excessive burden. Teams exploring research collaboration options for academic institutions may find this especially relevant when they need structured cognitive or behavioural assessments that fit real clinic workflows.
That practical layer matters in Montreal. Traffic, winter weather, caregiver availability, and language preference can all affect retention. A strong site plans for those realities.
CHUM and specialised institutes across the city
The Centre hospitalier de l'Université de Montréal (CHUM) is also central to the city’s research activity, with a strong presence in areas such as cardiology, cancer care, and metabolic disease. Its francophone and bilingual environment is part of what makes Montreal distinct. Communication, consent, and follow-up need to work in the language the participant understands best.
Several specialised centres add depth to the city’s research system. The Montreal Heart Institute is a well-known destination for cardiovascular studies. CHU Sainte-Justine Research Center is a major site for paediatric and maternal health research. The Montreal Neurological Institute-Hospital, often called The Neuro, remains a familiar name in neuroscience and neurology-related studies.
These centres do not all solve the same problem. A paediatric trial, a heart study, and a memory-related protocol require different staff, equipment, timelines, and family supports. That is why matching the participant to the right setting matters as much as finding an open trial.
How patients should think about these hubs
You do not need to memorize Montreal’s institutional map. You need a practical way to use it.
Start by matching the health problem to the likely type of centre, then ask one layer deeper. Where will the visits happen? Who is the study contact person? Can some assessments be done closer to home, by phone, or with digital tools? In cognitive, psychiatric, and neurological studies, those questions are especially important because repeated in-person testing can be tiring and can increase missing data if the process is too demanding.
A simple starting point looks like this:
Cancer care: ask whether the oncology team is connected to JGH, MUHC, CHUM, or another specialised cancer program.
Paediatric or maternal concerns: ask whether the study should be assessed through Sainte-Justine or another children’s research setting.
Heart and vascular conditions: ask whether a cardiovascular centre such as the Montreal Heart Institute is the better fit.
Brain, behaviour, or cognition questions: ask whether the study sits in neurology, psychiatry, rehabilitation, memory clinic, or developmental paediatrics, and whether modern cognitive assessment tools can reduce extra travel.
Complex chronic illness: ask whether a university-affiliated centre is better equipped because several specialists may need to coordinate care and trial procedures.
The strongest site for a trial is the one that fits the condition, the language of care, the visit burden, and the participant’s day-to-day reality.
How to Find and Evaluate Clinical Trials
A Montreal family often reaches the same moment. A specialist mentions a possible study, someone opens a laptop that evening, and within ten minutes the search turns confusing. One listing says recruiting. Another says active, not recruiting. A third sounds promising but gives no clear sense of how many visits are involved or whether the testing has to happen downtown.
That confusion is normal. Trial registries are built to document studies, not to guide people through real-life decisions about buses, parking, missed work, fatigue, childcare, or repeated cognitive testing.

Start with a search that matches real life
The best search is usually a narrow one. A registry works like a library catalogue. If you type only a broad topic, you get shelves of material. If you search with the right details, you get a short list worth reviewing.
Start with four filters, in this order:
Condition or symptom cluster
Use the clearest medical term you have. Examples include “mild cognitive impairment,” “major depressive disorder,” “migraine,” or a rare disease name from a specialist letter.Geography Add “Montreal,” “Quebec,” or a hospital name, as a study can be open in Canada but not open at a site you can realistically reach.
Recruitment status
Look for terms such as recruiting, enrolling, or active. If a listing is active but not recruiting, it may no longer be taking new participants.Study phase or study type
Some patients want later-stage studies with more prior experience behind them. Others are open to earlier-phase work if close monitoring and access to a new approach make sense for their situation.
If you work in a clinic and need concise background material before discussing options with a family, these clinical knowledge summaries can help support those conversations.
Read the listing like a screening tool
A trial listing is less like an advertisement and more like a triage note. It will not answer everything, but it usually tells you whether a follow-up call is worthwhile.
In September 2025, Canada reported thousands of active Phase I to IV trials. That historical snapshot matters less than the habit it points to. Use registries to create a shortlist, then verify the details with the local study team.
Here is how to read the common fields:
Listing detail | What it usually tells you |
|---|---|
Recruitment status | Whether the site may still be screening new participants |
Phase | How far along the intervention is in testing |
Inclusion and exclusion criteria | Who the study is designed for, and who may be ruled out for safety or scientific reasons |
Study location | Where visits may happen, which is not always the same as the city named in the title |
Primary outcome | What the researchers are actually measuring |
Contact information | Whether there is a clear path to ask questions or request screening |
Two lines deserve extra attention.
First, eligibility criteria. People often read these too quickly. A trial may fit the right diagnosis but exclude participants based on prior treatment, recent medication changes, kidney function, language of testing, or other health conditions.
Second, primary outcome. If a study focuses on a lab value, imaging marker, or cognitive score, that tells you what the team will be watching closely. It also hints at the burden of participation. A trial centered on repeated memory testing, mood scales, or neurologic exams may require several structured assessments over time.
Questions that help patients and clinicians quickly sort a trial
A short call or message to the study team should answer practical questions, not just scientific ones. In Montreal, logistics often decide whether a study is feasible.
Ask these early:
How many in-person visits are expected, and over what time period?
Which visits must happen at the main site?
Are any follow-ups possible by phone or video?
Are blood tests, questionnaires, or cognitive assessments repeated often?
Is testing offered in French, English, or both?
What happens if symptoms flare or transportation becomes difficult?
Is there reimbursement for parking, taxis, or transit?
For cognitive, psychiatric, and neurologic studies, repeated testing deserves special scrutiny. A protocol can look reasonable on paper and still create drop-off if participants must travel frequently for long assessments. Modern digital tools can help here by allowing some monitoring to happen with less disruption, which can reduce missing data and make participation more realistic for patients and families.
Study teams also need a dependable way to contact participants about visit reminders, symptom checks, and scheduling changes. Clear, secure outreach supports retention. Clinics comparing systems often review options for HIPAA compliant patient communication when building those workflows.
Decide whether the trial is plausible, not perfect
Perfection is the wrong standard at this stage. The useful question is whether the study deserves a proper conversation with your treating team.
A trial is usually worth a follow-up when three things line up. The diagnosis fits. The visit burden seems manageable. The study question matches a real clinical goal for the patient.
Send the listing to the clinician who knows the case best and ask one focused question: “Does this look reasonable for me, given my history and current treatment?”
That step saves time. A clinician may spot a mismatch in minutes, such as a recent medication change, a required prior therapy, an exclusion related to another illness, or an outcome measure that does not match the patient’s main concern.
For researchers, the lesson is just as practical. The easier a listing is to understand, the better the initial fit will be. Clear eligibility language, realistic descriptions of visit burden, and honest notes about remote versus in-person assessments help the right participants come forward.
Navigating Participation A Patient and Family Guide
Joining a trial rarely begins with a signature. It begins with a conversation.
A patient receives a call from a coordinator. A parent reads an email from a study nurse. An older adult brings a daughter or son to the first meeting because there’s a lot to absorb. That first contact is not a test. It’s the start of deciding whether the study fits your life, your goals, and your limits.

What informed consent really means
Informed consent is not just a form. It is the process of making sure you understand what the study is, what will happen, what the risks are, and what other options exist.
You should expect a clear explanation of:
Why the study is being done
What procedures are research-only
What side effects or burdens are known
Whether you might receive standard treatment, placebo, or an investigational intervention
How your privacy and study data will be protected
How to leave the study if you change your mind
A good consent discussion allows pauses, questions, and time to think. If the team rushes you, ask for the consent form to review at home. Bring a family member. Mark it up with questions.
The practical questions people forget to ask
The most common regrets I hear from participants are not about the science. They’re about logistics.
Some Montreal sites openly warn participants about construction and access issues, and compensation for some studies can range from $700 to $5,000, often not because of risk but to reflect time, travel, and multiple visits, according to Altasciences participant study information.
That means practical questions matter:
Travel support: Is parking reimbursed? Is public transit realistic from where I live?
Scheduling: Are early morning, evening, or school-friendly appointments available?
Visit length: Is this a quick check-in or half a day on site?
Caregiver role: Can someone accompany me? Will they need to stay the whole time?
Compensation timing: When and how is compensation provided?
Accessibility: Is the building easy for wheelchairs, walkers, fatigue, or sensory sensitivity?
Ask the coordinator to walk you through a typical study week, not just the ideal one.
That question often reveals the true burden. A study may sound manageable until you learn it requires fasting, traffic-heavy arrival windows, repeated forms, or multiple same-week visits.
Communication makes participation easier
Trial participation becomes much smoother when the study team communicates clearly between visits. Families often do better when reminders, changes, and follow-up instructions are organised and documented. If your institution is reviewing how teams should message participants safely, this guide to HIPAA compliant patient communication offers useful principles for secure outreach and reminder systems.
For families looking for support around cognition, learning, or next-step questions outside the trial itself, this resource for parents and individuals may also be helpful.
A simple decision framework for families
Not every eligible study is a good fit. I usually suggest weighing three things together.
Question | Why it matters |
|---|---|
Does this study match our goals? | Some people want access to new treatment. Others want close monitoring or to contribute to research. |
Can we realistically manage the visits? | Missed visits create stress for both the participant and the site. |
Do we trust the team enough to ask hard questions? | If you can’t speak openly before enrolment, participation may feel harder later. |
If the answer to the first question is yes, but the second is no, the study may still be the wrong choice for now. That is not failure. It is good judgement.
Enhancing Research with Modern Cognitive Assessment
Many clinical trials depend on outcomes that are hard to measure consistently. That problem shows up clearly in neurology, psychiatry, paediatrics, rehabilitation, and ageing research, where cognition often matters but isn’t always captured in a structured way.
A clinician may observe that a patient seems more attentive. A caregiver may report fewer daily mistakes. A therapist may notice better task completion. All of that is useful, but trials work best when those impressions are paired with repeatable data.
Why cognition is difficult to measure well
Traditional cognitive assessment can be valuable, but in research settings it often creates friction. It may require trained staff, fixed schedules, paper-heavy workflows, and long administration times. Participants may become tired, bored, or inconsistent across visits. Sites may struggle to standardise delivery across coordinators or across multiple locations.
Those practical limitations matter because recruitment and retention are already difficult. According to the North America clinical trials site analysis published by Credevo, patient recruitment and retention are major hurdles in 34% of North American trials. The same source notes that integrating real-time AI analytics, including 30-minute cognitive profiles, can provide actionable decision support and reduce costs by 15% to 25% through predictive retention models.
That doesn’t mean technology solves everything. It does mean the old approach of “we’ll measure cognition if we have time” is increasingly weak.
Better endpoints don’t just help the statistician. They help the coordinator know who is struggling, the investigator spot change earlier, and the participant stay engaged.
Where modern tools fit in Montreal research
Montreal has the right conditions for more advanced cognitive measurement. It has neurology services, psychiatry research, rehabilitation programmes, paediatric centres, memory clinics, and bilingual populations. What’s often missing is a practical bridge between those settings and scalable, low-friction assessment.
That’s where modern digital platforms become useful. In research, the ideal cognitive tool should be:
Brief enough to fit into study visits
Repeatable across time points
Engaging enough that participants will complete it
Structured enough to support protocol consistency
Flexible enough for adult and child-friendly workflows
Ethically deployable as a non-diagnostic research support tool when approved by the institution
For researchers designing CNS or neuropsychiatric studies, these Montreal cognitive assessment instructions show what an operational approach can look like when cognitive measurement needs to be practical rather than theoretical.
Why game-based and AI-supported methods are gaining attention
Participants often tolerate engaging, digitally delivered tasks better than repetitive paper tools, especially when repeat visits are required. That matters in long studies, paediatric work, and rehabilitation settings where motivation can fluctuate.
AI-supported analysis can also help sites identify patterns that manual review might miss. For example, changes in attention, executive function, or processing style across visits may signal adherence issues, fatigue effects, or the need for closer participant support. Used properly, this kind of signal can improve study operations rather than generating more data.
There’s also a broader care trend here. Outside formal trials, digital mental health and self-management tools are becoming more common, and something like the lunabloomai app reflects how participants are getting used to structured digital support in health contexts. Research teams can learn from that expectation. People increasingly want tools that are responsive, understandable, and less burdensome.
Practical examples for different research teams
A few examples make this more concrete.
In a memory-related study, the team may need repeat measures of attention and memory without adding a long neuropsychology battery at every visit. A brief digital cognitive protocol can support trend monitoring between larger formal assessments.
In a paediatric neurodevelopmental trial, child engagement is often half the battle. A game-based format may reduce resistance and improve completion, especially when parents already feel overloaded by forms and appointments.
In psychiatry research, symptom scores remain important, but they don’t always show how a patient is functioning cognitively. Structured tasks can add another layer of evidence when treatment effects on concentration or executive function matter.
In rehabilitation studies, repeated measurement can help distinguish perceived improvement from observable task change. That is useful when clinicians want to know whether progress is consistent, not just whether a participant had a good day.
What researchers should ask before adopting a cognitive platform
Not every digital tool belongs in a trial. Before adding one, ask:
Is it aligned with the protocol endpoint or exploratory objective?
Can it be administered consistently across sites and staff?
Does the participant experience support completion rather than create fatigue?
Can the output be reviewed in a way that helps decision-making during the study?
Has the ethics team approved its role and data flow?
If the answer to those questions is yes, modern cognitive assessment can move from being an afterthought to becoming one of the most useful parts of the study design.
Your Next Step in Montreal's Research Future
Montreal offers something rare. It has major hospitals, specialised research units, active trial infrastructure, and a patient population that makes serious clinical research possible. For patients and families, that means there are real places to ask for help. For clinicians, it means referral options often exist even when standard care has stalled. For researchers, it means the ecosystem is strong enough to support better study design.
The challenge is not only finding studies. It’s making them workable.
Patients need plain-language explanations, realistic scheduling, and honest discussions about travel, consent, and support. Researchers need cleaner workflows, stronger retention, and outcomes that reflect how people function. Clinicians need tools that help them judge whether a study fits a specific person sitting in front of them.
One unmet need stands out clearly. An overview of Montreal trial listings and gaps points to limited detail on how modern cognitive assessment tools can be integrated, despite a clear need in mild cognitive impairment and neurodevelopmental studies. That gap matters because cognition is often central to patient experience, yet still undermeasured or inconsistently measured in many protocols.
Montreal’s research future won’t be shaped only by new drugs or new devices. It will also be shaped by better measurement, better participant support, and better coordination between hospitals, families, and research teams.
If you’re exploring clinical trials in montreal, start with one practical action. Ask your clinician about a specific study. Contact a site coordinator with focused questions. Review whether the visit burden fits your life. If you’re building a study, look closely at where recruitment, retention, and outcome measurement are likely to fail, then improve those parts first.
If you’re a clinician, researcher, academic team, or family looking to strengthen brain health research and cognitive measurement in Quebec, visit Orange Neurosciences or contact the team to explore collaboration, ethical research integration, and practical tools for cognitive assessment in Montreal and beyond.

Orange Neurosciences' Cognitive Skills Assessments (CSA) are intended as an aid for assessing the cognitive well-being of an individual. In a clinical setting, the CSA results (when interpreted by a qualified healthcare provider) may be used as an aid in determining whether further cognitive evaluation is needed. Orange Neurosciences' brain training programs are designed to promote and encourage overall cognitive health. Orange Neurosciences does not offer any medical diagnosis or treatment of any medical disease or condition. Orange Neurosciences products may also be used for research purposes for any range of cognition-related assessments. If used for research purposes, all use of the product must comply with the appropriate human subjects' procedures as they exist within the researcher's institution and will be the researcher's responsibility. All such human subject protections shall be under the provisions of all applicable sections of the Code of Federal Regulations.
© 2026 by Orange Neurosciences Corporation


